- Key Takeaways
- Part 1. What is a licoo₂ battery?
- Part 2. Licoo₂ battery reaction: how it works
- Part 3. Advantages and limitations of lithium cobalt oxide batteries
- Part 4. The cobalt challenge and industry response
- Part 5. Typical applications of licoo₂ batteries
- Part 6. Licoo₂ vs other lithium-ion chemistries
- Part 7. How to extend licoo₂ battery life
- Part 8. Licoo₂ battery FAQs
LiCoO₂ (commonly searched as “licoo2 battery” or lithium cobalt oxide battery) is one of the earliest and most established lithium-ion cathode chemistries. With a practical energy density of 150–200 Wh/kg and a stable 3.7 V nominal voltage, LiCoO₂ batteries remain a core solution for compact, high-energy electronic systems.
This guide explains what a LiCoO₂ battery is, how the LiCoO₂ battery reaction works, where this chemistry is still used today, and how engineers should evaluate it against LiFePO₄ and NMC options in real-world selection scenarios.
Key Takeaways
- LiCoO₂ batteries prioritize high energy density and compact size, not extreme cycle life or abuse tolerance.
- The LiCoO₂ battery reaction relies on reversible lithium intercalation and requires strict voltage control (≤ 4.2 V).
- Compared with LiFePO₄, lithium cobalt oxide offers higher energy density but lower thermal stability.
- Cobalt cost and sourcing risk are the main long-term constraints of LiCoO₂ chemistry.
- Best suited for consumer electronics, medical devices, and precision instruments.
Part 1. What is a licoo₂ battery?
A LiCoO₂ battery is a rechargeable lithium-ion battery that uses lithium cobalt oxide (LiCoO₂) as the cathode and graphite as the anode. First commercialized by Sony in the early 1990s, this chemistry became the technical foundation of modern lithium-ion batteries.
Defining Characteristics
- High gravimetric energy density for space-constrained designs
- Stable discharge voltage around 3.7 V
- Mature manufacturing ecosystem with predictable performance and aging behavior
Although electric vehicles now favor NMC or LFP chemistries, LiCoO₂ remains dominant in consumer electronics and selected medical and industrial applications where size and weight are critical.
Part 2. Licoo₂ battery reaction: how it works
Understanding the LiCoO₂ battery reaction is essential for safe system design and lifecycle optimization.
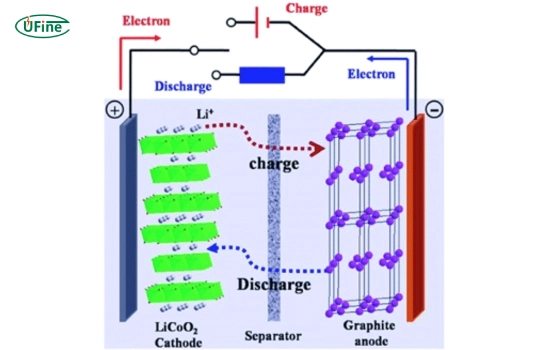
1 Electrochemical principle
During charging, lithium ions are de-intercalated from the LiCoO₂ cathode and migrate through the electrolyte to the graphite anode. During discharge, lithium ions return to the cathode, releasing electrical energy.
Simplified reactions:
- Charge: LiCoO₂ → Li₁₋ₓCoO₂ + xLi⁺ + xe⁻
- Discharge: Li₁₋ₓCoO₂ + xLi⁺ + xe⁻ → LiCoO₂
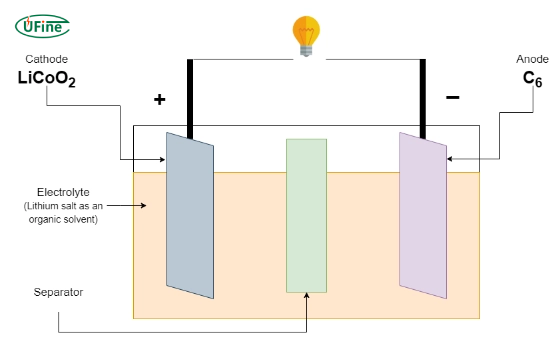
2 Core cell components
- Cathode: Lithium cobalt oxide (LiCoO₂)
- Anode: Graphite (C₆)
- Electrolyte: Lithium salt (commonly LiPF₆) in organic solvents
- Separator: Microporous polymer preventing internal short circuits
Engineering note: Delithiation beyond ~50% destabilizes the LiCoO₂ crystal lattice. This is why 4.2 V maximum charge voltage and a reliable BMS are mandatory.
Part 3. Advantages and limitations of lithium cobalt oxide batteries
1 Advantages
- High Energy Density: Ideal for compact, lightweight devices
- Stable Voltage Output: Suitable for sensitive electronics
- Mature Supply Chain: Consistent quality and predictable degradation
- Good Manufacturability: High yield and uniform cell behavior
2 Limitations
- Thermal Stability: Lower safety margin than LiFePO₄; protection circuits required
- Cycle Life: Typically 500–1,500 cycles, depending on operating conditions
- Cobalt Dependency: Higher cost and ESG concerns
- Moderate Power Capability: Not optimized for high-C-rate or fast charging
Part 4. The cobalt challenge and industry response
Cobalt sourcing is a major economic and regulatory issue. A large share of global cobalt supply originates from the DRC, creating cost volatility and ESG pressure.
Industry responses include:
- Reduced-cobalt chemistries: NMC and NCA with lower cobalt content
- Closed-loop recycling: Hydrometallurgical recovery exceeding 90%
- Material innovation: Solid-state and cobalt-free cathode research
For sustainability context, refer to the International Energy Agency report on critical minerals in clean energy transitions.
Part 5. Typical applications of licoo₂ batteries
LiCoO₂ batteries are best suited for applications prioritizing energy density over cycle life:
- Consumer Electronics: Smartphones, laptops, tablets, wearables
- Medical Devices: Infusion pumps, portable diagnostic equipment
- Industrial Instruments: Handheld analyzers, precision test tools
- Legacy EV Systems: Early-generation traction or auxiliary packs
For long-life and safety-critical systems, see our internal comparison with LiFePO₄ battery technology.
Part 6. Licoo₂ vs other lithium-ion chemistries
1 Technical comparison (2025 benchmark)
| Feature | LiCoO₂ | LiFePO₄ | NMC 811 |
|---|---|---|---|
| Energy Density | 150–200 Wh/kg | 90–120 Wh/kg | 220–280 Wh/kg |
| Cycle Life | 500–1,500 | 2,000+ | 800–1,200 |
| Thermal Stability | Moderate | High | Moderate |
| Cost | High | Moderate | High |
| Typical Use | Compact electronics | ESS, backup power | EV traction |
2 Selection guidance
- Choose LiCoO₂ when space and weight dominate design constraints
- Choose LiFePO₄ for long life and safety-critical systems
- Choose NMC for high-energy traction and industrial platforms
Part 7. How to extend licoo₂ battery life
From an engineering perspective, degradation is driven mainly by voltage stress and temperature.
Best practices:
- Limit charging voltage to ≤ 4.2 V
- Operate between 10 °C and 35 °C when possible
- Store at 40–60% SOC for long idle periods
- Avoid deep discharge below 3.0 V
- Use a calibrated BMS with accurate cell balancing
Part 8. Licoo₂ battery FAQs
What is the typical lifespan of a LiCoO₂ battery?
Most LiCoO₂ batteries deliver 500–1,500 cycles, depending on depth of discharge, temperature, and charge voltage.
Are LiCoO₂ batteries still used in electric vehicles?
They are largely phased out of modern EV packs but remain relevant in high-energy auxiliary or legacy systems.
What voltage does a LiCoO₂ battery operate at?
Nominal voltage is 3.7 V, with an operating range of 3.0–4.2 V.
How can I tell if a LiCoO₂ battery is degrading?
Common signs include capacity dropping below 80%, abnormal heating, swelling, or rising internal resistance.
Is a lithium cobalt oxide battery safe?
Yes, when properly designed with a BMS and thermal protection. Without protection, LiCoO₂ has a lower safety margin than LFP.
Related Tags:
More Articles

A Complete Guide to 12V Batteries and Their Types
Learn what a 12V battery is, explore types of 12V batteries, and compare lead-acid vs lithium. Clear guide for selection, applications, and performance.
How to Increase 18650 Battery Life (Practical Guide + Cycle Life Calculation)
Learn how to extend 18650 battery life with proper charging, storage, and usage. Includes 18650 battery life cycle calculation and expert tips.
What Is a Lithium Titanate Battery?
Discover lithium titanate batteries (LTO), including how they work, key benefits, and real-world uses. Compare LTO vs lithium-ion and lead-acid batteries.
How Manufacturers Can Tell You About the Cost of Lithium Batteries
Learn real lithium battery pricing, cost per kWh, and how manufacturers set prices using materials, chemistry, and production scale.
How Long Does A Drone Battery Last?
Learn drone battery flight time, lifespan cycles, and tips to extend performance. Includes drone battery price and Mavic Air battery insights.