- Key takeaways
- Part 1. What are zinc batteries?
- Part 2. How do zinc-based batteries work?
- Part 3. Pros and cons of zinc-based batteries
- Part 4. How zinc‑ion differs from lithium‑ion (and other zinc chemistries)
- Part 5. Materials and how chemistry shapes performance
- Part 6. Where zinc batteries actually shine
- Part 7. Performance comparison: zinc vs lithium in real scenarios
- Part 8. Cost and economic perspective
- Part 9. Real-life scenarios and user insights
- Part 10. FAQs
Key takeaways
- Zinc‑based batteries are emerging as safer and more sustainable alternatives to lithium‑ion, especially for stationary storage and affordable energy systems.
- You’ll see clear trade‑offs: lower energy density than Li‑ion but higher safety, material abundance, and recyclability.
- Zinc‑ion, zinc‑lithium, and zinc‑air are related but distinct chemistries with their own strengths and real‑world niches.
- Technical challenges like dendrite growth, electrolyte stability, and cycle life limits are key R&D focus areas.
- Practical applications today are centered more on grid storage, backup power, and safe consumer products — not yet fully replacing lithium in EVs.
Part 1. What are zinc batteries?
Zinc-based batteries are a category of electrochemical energy storage devices that use zinc as a primary component in their electrodes. Known for their simplicity, affordability, and safety, these batteries have been around for decades but are now gaining renewed attention as advancements address historical limitations.
There are two main types of zinc-based batteries: zinc-air batteries and zinc-ion batteries. Both leverage zinc’s natural properties—high energy density, abundance, and non-toxicity—to deliver power. Unlike lithium, which is rare and requires complex mining processes, zinc is the 24th most abundant element in Earth’s crust, making it a sustainable choice for large-scale applications.
Part 2. How do zinc-based batteries work?
1 Understanding zinc-air batteries
Zinc-air batteries are best known for their high energy density—up to five times greater than lithium-ion batteries in some cases. This makes them ideal for applications where space and weight matter, such as hearing aids, military equipment, and electric vehicles (EVs).
However, traditional zinc-air batteries have limitations:
-
They are typically primary batteries (non-rechargeable), though research into rechargeable versions is advancing.
-
Exposure to air can cause electrolyte evaporation, reducing lifespan.
Recent innovations, such as improved air cathodes and hybrid electrolytes, are addressing these challenges, making zinc-air batteries a contender for grid storage and EV markets.
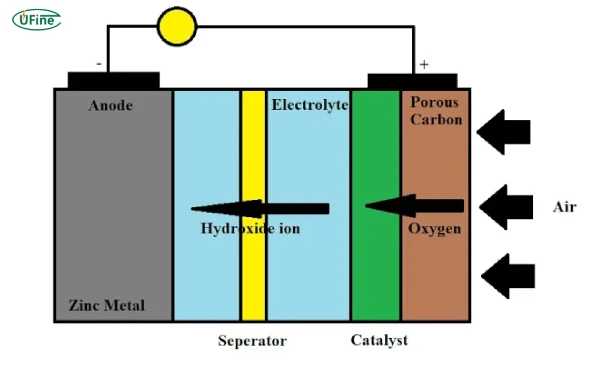
How Do Zinc-Air Batteries Work
Zinc-air batteries generate electricity through a reaction between zinc and oxygen from the air. Here’s a simplified breakdown:
-
Anode: Made of zinc metal.
-
Cathode: A porous electrode that allows oxygen from the air to react.
-
Electrolyte: Typically an alkaline solution (e.g., potassium hydroxide).
When the battery discharges, zinc at the anode oxidizes, releasing electrons. Oxygen from the air reacts with water and electrons at the cathode, forming hydroxide ions. These ions travel through the electrolyte to complete the circuit, producing electricity.
2 Understanding zinc-ion batteries
Zinc-ion batteries are emerging as a rechargeable alternative to lithium-ion systems. Key features include:
-
Water-based electrolytes: Unlike flammable organic electrolytes in lithium-ion batteries, zinc-ion systems use safer aqueous solutions.
-
High cycle life: Some prototypes achieve over 1,000 charge-discharge cycles.
-
Low cost: Zinc is cheaper than lithium, cobalt, or nickel.
These batteries are being tested for use in renewable energy storage, consumer electronics, and even electric scooters. Companies like Eos Energy Enterprises and NantEnergy are commercializing zinc-ion technology for large-scale applications.
How Do Zinc-Ion Batteries Work
Zinc-ion batteries operate similarly to lithium-ion batteries but use zinc ions (Zn²⁺) instead of lithium ions. During discharge:
-
Zinc ions move from the anode (zinc metal) through the electrolyte to the cathode (often a manganese oxide or vanadium-based material).
-
Electrons flow through an external circuit, powering devices.
-
During charging, the process reverses, with zinc ions returning to the anode.
Part 3. Pros and cons of zinc-based batteries
Advantages
-
Safety: No risk of thermal runaway or fires (common in lithium-ion batteries).
-
Sustainability: Zinc is abundant, recyclable, and less environmentally damaging to extract.
-
Cost-Effective: Raw materials are cheaper, and manufacturing processes are simpler.
-
High Energy Density: Especially true for zinc-air variants.
Disadvantages
-
Lower Voltage: Zinc-based batteries typically deliver 1.2–1.6V, compared to lithium-ion’s 3.6V.
-
Limited Cycle Life: Rechargeable versions still lag behind lithium-ion in longevity.
-
Air Sensitivity: Zinc-air batteries require careful sealing to prevent electrolyte degradation.
Part 4. How zinc‑ion differs from lithium‑ion (and other zinc chemistries)
Let’s compare the big players. You’ll notice zinc batteries don’t yet dominate EV markets, and that’s for good reasons that go beyond marketing hype:
| metric | lithium‑ion battery | zinc‑ion battery |
|---|---|---|
| energy density | relatively high (≈150–250 Wh/kg) | lower (≈60–120 Wh/kg) |
| cycle life | 500–3000+ cycles | potentially >3000 cycles when optimized |
| safety | risk of thermal runaway | high safety, low fire risk |
| raw material cost | higher, supply risk | lower, abundant zinc |
| electrolyte | organic solvents | mainly aqueous (safer) |
In contrast with true zinc‑ion batteries — which use Zn²⁺ shuttling in intercalation cathodes — zinc‑air batteries use oxygen from air and tend to aim for very high theoretical energy density but historically suffer from rechargeability issues. Meanwhile, something like a “zinc‑lithium battery” may combine chemistries to try and boost voltage or energy characteristics, but it’s a different beast altogether from a pure Zn‑ion cell.
Part 5. Materials and how chemistry shapes performance
What makes zinc batteries both promising and tricky is their materials palette. Let’s talk about that:
1. anode (negative side)
- Usually metallic zinc, which gives a theoretical capacity of ~820 mAh/g — quite respectable.
- But zinc plating/stripping cycles can grow dendrites — tiny needle‑like structures that can short‑circuit a cell if uncontrolled.
2. cathode (positive side)
- Typical zinc‑ion cathodes are manganese oxides or vanadium‑based materials that accept Zn²⁺ ions during discharge.
- Engineers are actively optimizing these to improve capacity retention and voltage stability.
3. electrolyte
- Unlike Li‑ion (which relies on flammable organic liquids), many Zn batteries use water‑based electrolytes for enhanced safety.
- However, at high voltages, water decomposition and hydrogen evolution can occur — so electrolyte chemistry is a hot R&D area.
The result? You gain safety and cost benefits but still wrestle with achieving high energy density and stable cycling comparable to Li‑ion.
Part 6. Where zinc batteries actually shine
Here’s the part where zinc tech feels practical — and where you can start imagining real applications:
stationary energy storage (renewables + grid)
- Zinc‑ion batteries are being looked at for grid or solar storage because cost per kWh and safety matter more than pack weight.
- The use of aqueous electrolytes significantly reduces the risk of fire, making them more suitable for installations in homes and utility facilities.
backup power & UPS systems
- Because of their long life and stable chemistry, these batteries can serve as reliable backup power units.
safety‑critical consumer electronics
- Devices where overheating or combustion is unacceptable (medical, industrial sensors, etc.) may benefit from zinc‑based chemistries.
Meanwhile, for electric vehicles, the higher energy density and faster charge capabilities of advanced lithium chemistries still offer a compelling edge — but zinc is closing gaps in safety and cost.
Part 7. Performance comparison: zinc vs lithium in real scenarios
| Metric | Lithium-ion | Zinc-ion | Zinc-lithium | Real-world use case |
|---|---|---|---|---|
| Energy density | 150–250 Wh/kg | 60–120 Wh/kg | 100–150 Wh/kg | EV: Li-ion; home storage: Zn-ion |
| Cycle life | 500–3000+ | >3000 (optimized) | ~2000 | Longer life reduces replacement cost |
| Safety | Moderate risk of thermal runaway | High safety | High safety | Indoor applications, sensitive environments |
| Cost per kWh | High | Lower | Moderate | Budget-conscious storage solutions |
| Electrolyte | Organic solvents | Aqueous | Mixed | Safety vs performance trade-off |
You’ll notice the trade-offs: lithium dominates EVs for high energy density, but zinc batteries are perfect for backup power, solar storage, and safety-critical devices. If you imagine a hospital UPS system or a home battery in a hot attic, you’d probably prefer zinc over lithium — the peace of mind is worth slightly lower energy density.
Part 8. Cost and economic perspective
Let’s talk money — because it matters.
- Raw material costs: Zinc is cheap and abundant, whereas lithium prices can swing wildly due to supply shortages.
- Lifecycle cost: Because zinc-ion batteries often outlast standard lithium packs in stationary storage, your cost per kWh over 10–15 years can be lower.
- Recycling and environmental impact: Zinc is non-toxic, and recycling processes are simpler than for Li-ion, which appeals to eco-conscious users.
Imagine you’re comparing two systems: a lithium storage unit and a zinc-ion storage unit of the same usable capacity. Even if the upfront cost is similar, the zinc battery’s longer safe lifespan, lower replacement frequency, and reduced fire insurance costs may make it more economical in the long run.
Part 9. Real-life scenarios and user insights
- Home energy storage: Safe indoors, predictable performance, compatible with solar setups.
- Backup power / UPS: Long cycle life and low fire risk make zinc-ion ideal for hospitals or industrial systems.
- Portable or safety-critical devices: Medical devices, sensors, or consumer electronics benefit from low flammability.
You can see a pattern: where safety, stability, and longevity matter more than raw energy density, zinc wins. Conversely, if you’re packing a high-performance EV battery, lithium still holds the crown.
Part 10. FAQs
How does temperature affect zinc battery performance?
Zinc batteries handle moderate heat well, but extreme cold can slow ionic movement, reducing output temporarily.
Can you mix zinc and lithium batteries in the same system?
No — they have different charging profiles and voltage behavior. Mixing can cause uneven charging and damage.
How fast can zinc batteries be charged compared to lithium?
Zinc-ion generally charges slower than lithium-ion at high currents, but optimized cells can reach reasonable fast-charge rates safely.
What maintenance is required for zinc batteries?
Minimal — keep them in moderate temperatures, avoid deep over-discharge, and check electrolyte levels if applicable.
Are zinc batteries prone to memory effect?
Not significantly. They tolerate partial discharges better than older Ni-Cd cells.
Can zinc batteries be used in off-grid solar setups?
Absolutely — they’re ideal for solar storage due to safety, low maintenance, and long cycle life.
Related Tags:
More Articles

The Ultimate Guide to Small Size Battery
Learn small battery types, sizes, and applications. Compare lithium, alkaline, and coin cells. Find the best small size battery for your device or project.
Lithium Battery Comparison: Soft Pack Vs. Hard Pack
Compare soft pack vs hard pack lithium battery. Understand structure, safety, energy density, and applications to choose the right battery.
What Size are 18650 Batteries?
Learn exact 18650 battery dimensions (18mm × 65mm), tolerances, and pack configurations. Compare 18650 size with 21700 and others for better battery selection.
The Ultimate Guide to Battery Terminal Clamps
Learn battery terminal clamps, including types, materials, installation, and how to choose the right battery clamp for car and industrial use.
What Is a Lithium-Ion Battery Pack?
Learn what a lithium ion battery pack is, how it works, key components, configurations, and real-world applications for industrial and commercial use.