- Key takeaways
- Part 1. What is lithium oxidation
- Part 2. What is battery oxidation
- Part 3. What causes lithium battery oxidation?
- Part 4. How lithium oxidation affects battery performance
- Part 5. Lithium battery oxygen signal
- Part 6. Can oxidized lithium batteries still be used?
- Part 7. How to reduce lithium oxidation in batteries
- Part 8. FAQs
Lithium is one of the most reactive metals you can use in energy storage. That reactivity is exactly why lithium batteries deliver such high energy density — but it’s also why lithium oxidation becomes an important topic if you care about performance, safety, and long-term reliability.
If you’ve ever noticed battery capacity slowly fading, internal resistance rising, or batteries aging faster than expected, oxidation is often part of the story.
In this guide, you’ll learn what lithium oxidation actually means, whether lithium is an oxidizer, how oxidation affects lithium batteries, and — most importantly — what you can do about it.
Key takeaways
- Lithium oxidizes very easily when exposed to oxygen or moisture
- Lithium itself is not an oxidizer — it acts as a strong reducing agent
- Lithium oxidation is closely related to battery aging and performance degradation
- Electrode oxidation and electrolyte oxidation are the two main battery oxidation mechanisms
- Battery manufacturers use coatings, additives, and sealing techniques to reduce oxidation
Part 1. What is lithium oxidation
Lithium oxidation is a chemical reaction in which lithium loses electrons and reacts with oxygen or other oxidizing substances. Because lithium is extremely reactive, this process can happen quickly — even under mild conditions.
When lithium metal reacts with oxygen, it typically forms lithium oxide:
4Li + O₂ → 2Li₂O
This reaction explains why lithium metal is usually stored in sealed containers or under mineral oil. If exposed to air, lithium begins oxidizing almost immediately.
In lithium batteries, oxidation doesn’t always happen in the same obvious way. Instead of visible oxidation like rust on iron, lithium battery oxidation often occurs inside the battery, gradually affecting performance.
1 Does lithium oxidize easily
Yes — lithium oxidizes very easily.
In fact, lithium is one of the most reactive metals in the periodic table. When lithium is exposed to:
- oxygen
- moisture
- carbon dioxide
- electrolytes
…it begins to oxidize or react chemically.
This high reactivity is both an advantage and a challenge. On one hand, it allows lithium batteries to store more energy. On the other hand, it also makes lithium batteries more sensitive to environmental and chemical changes.
For example, during battery cycling, small amounts of oxygen or electrolyte decomposition can trigger oxidation reactions at the electrode surface. Over time, these reactions accumulate and lead to measurable performance degradation.
So while lithium oxidation might sound like a purely chemical concept, it has very real consequences in battery applications.
2 Is lithium an oxidizer
This is a common question — and the answer often surprises people.
Lithium is not an oxidizer.
Instead, lithium is a reducing agent. That means lithium tends to lose electrons, not gain them.
Here’s the difference:
| Term | What it means | Lithium behavior |
|---|---|---|
| Oxidizer | Gains electrons | Lithium does not |
| Reducing agent | Loses electrons | Lithium acts this way |
Because lithium readily loses electrons, it drives oxidation reactions in other materials. In lithium batteries, this property enables electron flow — which is essential for generating electricity.
If you’re wondering why this matters, here’s the practical takeaway:
Lithium itself isn’t oxidizing other materials in the traditional sense — it’s participating in electrochemical reactions that can eventually lead to battery oxidation effects.
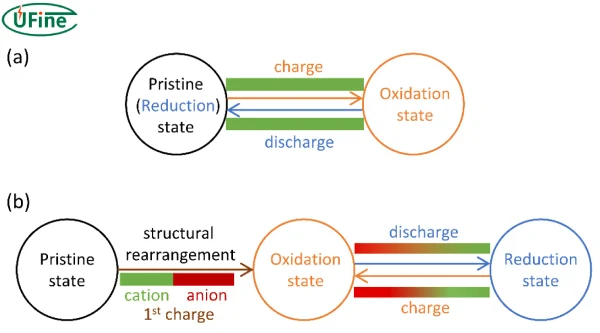
3 Oxidation state of lithium
Lithium has a very stable oxidation state of +1.
This is because lithium has one electron in its outer shell. When lithium loses that electron, it forms a stable ion:
Li → Li⁺ + e⁻
This +1 oxidation number is extremely important in lithium battery chemistry. It explains why lithium ions move between electrodes during charge and discharge cycles.
Here’s a quick reference:
| Property | Lithium |
|---|---|
| Atomic number | 3 |
| Valence electrons | 1 |
| Common oxidation state | +1 |
| Battery form | Li⁺ ion |
Because lithium strongly prefers this +1 oxidation state, lithium-ion batteries rely on reversible lithium ion movement instead of permanent chemical changes.
However, oxidation reactions inside the battery can still occur — and that’s where battery oxidation becomes important.
Part 2. What is battery oxidation
Lithium battery oxidation is a complex process involving chemical reactions within the battery. It primarily occurs when lithium within the battery reacts with oxygen, forming lithium oxide. This reaction can begin during the manufacturing process or develop over time as the battery is used.
The Principle of Lithium Oxidation
To understand lithium oxidation, it’s essential to grasp the underlying principles. Lithium, being highly reactive, tends to form compounds with other elements. When lithium comes into contact with oxygen, it readily reacts to form lithium oxide (Li2O). This reaction can be likened to the rusting of iron. Just as iron oxidizes to form rust when exposed to oxygen and moisture, lithium oxidizes to form lithium oxide under similar conditions.
Part 3. What causes lithium battery oxidation?
Understanding the causes of lithium battery oxidation can help in preventing this issue. Here are some common causes:
-
Exposure to Air: Lithium is highly reactive with oxygen. When a lithium battery is exposed to air, the lithium inside reacts with oxygen to form lithium oxide, leading to oxidation.
-
Moisture: Humid environments can accelerate the oxidation process. Water molecules can infiltrate the battery and react with lithium, forming lithium hydroxide, which further oxidizes to lithium oxide.
-
High Temperatures: Heat speeds up chemical reactions. High temperatures can accelerate the oxidation process, causing the battery to degrade faster.
-
Overcharging: Charging the battery beyond its recommended levels can lead to increased internal pressure and temperature, which promotes oxidation.
-
Aging: Over time, batteries naturally degrade. Older batteries are more prone to oxidation due to prolonged exposure to various environmental factors and the gradual breakdown of their internal components.
To better understand how oxidation contributes to long-term degradation, you can also read our guide on lithium battery aging.
Chemical Reactions Involved
The primary chemical reaction involved in lithium oxidation is: 4Li+O2→2Li2O
In the presence of moisture, the reaction can be: 2Li+2H2O→2LiOH+H2
Further oxidation leads to: 4LiOH+O2→2Li2O+2H2O
These reactions highlight how lithium interacts with oxygen and moisture, leading to the formation of lithium oxide and other compounds that contribute to battery degradation
Part 4. How lithium oxidation affects battery performance
Lithium oxidation doesn’t usually cause sudden failure. Instead, it creates gradual performance changes that you may notice over months or years.
Here’s how oxidation typically impacts batteries:
| Oxidation effect | What happens | Result |
|---|---|---|
| Electrode oxidation | Surface layer forms | Reduced capacity |
| Electrolyte oxidation | Decomposition products build up | Increased resistance |
| Structural degradation | Active material damage | Shorter lifespan |
| Gas generation | Chemical reactions | Battery swelling |
One of the most important oxidation-related phenomena is SEI layer formation (solid electrolyte interphase). This layer forms naturally during early battery cycles and helps stabilize the battery.
However, excessive oxidation can thicken the SEI layer, which slows lithium ion movement and reduces battery efficiency.
This is why battery engineers often talk about “aging” — in many cases, oxidation is a major contributor.
For technical definitions of oxidation and reduction, you can also refer to Encyclopedia Britannica – Oxidation-reduction reaction.
Part 5. Lithium battery oxygen signal

Identifying the signs of lithium battery oxidation early can help in mitigating its adverse effects. Here are some signals that indicate a lithium battery might be undergoing oxidation:
-
Swelling: One of the most noticeable signs of oxidation is the swelling or puffing up of the battery. This occurs because the formation of lithium oxide increases the internal pressure within the battery.
-
Heat: An oxidizing lithium battery may become unusually hot during charging or use. This excess heat is due to the exothermic nature of the oxidation reaction.
-
Reduced Performance: If you notice that the battery is not holding a charge as well as it used to or that it discharges more quickly, oxidation could be a contributing factor.
-
Discoloration: The battery casing or the terminals might show signs of discoloration. This is due to the chemical reactions occurring within the battery.
-
Odor: A chemical smell emanating from the battery can also indicate oxidation. This smell is often a result of the breakdown of the battery’s electrolyte.
Monitoring and Early Detection
Regularly monitoring your lithium batteries for these signals is crucial. Early detection of oxidation can prevent further damage and ensure the safety and longevity of your batteries. If you observe any of these signs, it’s essential to take immediate action.
Part 6. Can oxidized lithium batteries still be used?
While it is technically possible to use oxidized lithium batteries, it is not recommended. Here’s why:
-
Compromised Performance: Oxidized batteries do not perform as well as non-oxidized ones. Their reduced capacity and efficiency mean that they will not power devices for as long or as effectively.
-
Safety Risks: The safety hazards associated with oxidized batteries, such as swelling, leaks, and potential explosions, make their use risky. It’s better to replace them to avoid these dangers.
-
Inconsistent Reliability: Oxidized batteries may exhibit inconsistent performance, leading to sudden power drops or failures. This unreliability can be problematic, especially in critical applications.
When to Replace an Oxidized Battery
If you notice any signs of oxidation, such as swelling, heat, or reduced performance, it’s best to replace the battery. Using a compromised battery can lead to further damage to your device and pose safety risks.
Part 7. How to reduce lithium oxidation in batteries
Battery manufacturers use several techniques to control oxidation and improve battery life. Some of these strategies are invisible to users — but they make a significant difference.
For example, protective coatings can be applied to cathode materials to reduce oxidation at high voltages. Electrolyte additives can stabilize chemical reactions. And improved sealing prevents moisture from entering the battery.
At Ufine Battery, these techniques are carefully integrated into custom lithium battery designs to enhance long-term stability and performance.
Temperature management also plays a major role. When batteries operate at higher temperatures, oxidation reactions accelerate. Keeping batteries within recommended temperature ranges can significantly extend lifespan.
If you’re selecting batteries for a long-term project, these design differences matter. Two batteries may look identical on paper, but oxidation control technology can dramatically affect real-world performance.
For us ordinary users, here are some effective prevention tips:
-
Store Properly: Store batteries in a cool, dry place away from direct sunlight. Extreme temperatures and humidity can accelerate oxidation.
-
Avoid Overcharging: Follow the manufacturer’s charging guidelines. Overcharging can increase internal pressure and temperature, promoting oxidation.
-
Use Protective Casings: Protective casings can shield batteries from air and moisture, reducing the risk of oxidation.
-
Regular Checks: Inspect your batteries regularly for any signs of oxidation. Early detection can help prevent further damage.
-
Temperature Control: Avoid exposing batteries to high temperatures. Keeping them at a moderate temperature can slow down the oxidation process.
-
Keep Contacts Clean: Ensure that the battery contacts are clean and free from corrosion. Corroded contacts can increase resistance and generate heat, accelerating oxidation.
Best Practices for Battery Maintenance
Implementing best practices for battery maintenance can significantly reduce the risk of oxidation. Regularly check your batteries, follow proper charging and storage procedures, and replace batteries showing signs of wear or oxidation.
Part 8. FAQs
1. Can lithium oxidation cause battery swelling?
In some cases, yes. Oxidation-related reactions can produce gases inside the battery. When gas accumulates, it may cause battery swelling or deformation.
2. Is lithium oxidation dangerous?
Lithium oxidation itself is not always dangerous. However, excessive oxidation inside a battery can increase heat generation, which may raise safety risks under extreme conditions.
3. Does lithium oxidation happen during charging?
Yes. Oxidation reactions are more likely to occur during charging, especially at high voltages or fast charging conditions. This is why fast charging can sometimes accelerate battery aging.
4. Does temperature affect lithium oxidation?
Absolutely. Higher temperatures accelerate chemical reactions, including oxidation. Operating lithium batteries at elevated temperatures can significantly increase oxidation and reduce lifespan.
5. Do all lithium batteries experience oxidation?
Yes. All lithium batteries experience some level of oxidation over time. However, high-quality batteries are designed to minimize oxidation and maintain performance longer.
6. Can lithium oxidation be reversed?
No. Most lithium oxidation reactions are irreversible. Once oxidation occurs, the resulting performance loss cannot be fully recovered.
7. Which lithium batteries resist oxidation better?
Lithium iron phosphate (LiFePO4) batteries typically show better oxidation stability compared to high-energy lithium-ion chemistries, especially at higher temperatures and voltages.
Related Tags:
More Articles

What is UN38.3? Learn UN38.3 lithium battery transport tests, requirements, certification process, and compliance tips for safe global shipping.
Differences Between a Car Battery and a Truck Battery
Compare truck battery vs car battery differences, including CCA, capacity, size, and applications. Learn how to choose the right battery.
Top 10 Recommended Small Size Battery
Learn about small size batteries, including types, uses, and how to choose the right one. Compare options for different devices and applications.
Voltage vs Current: What is the Difference?
Learn voltage vs current, their differences, relationship, and battery applications. Clear examples explain current vs voltage and power.
Can You Overcharge a Lithium Battery?
Can you overcharge a lithium battery? Learn what happens, key risks, and how to prevent battery overcharging for safer and longer battery life.