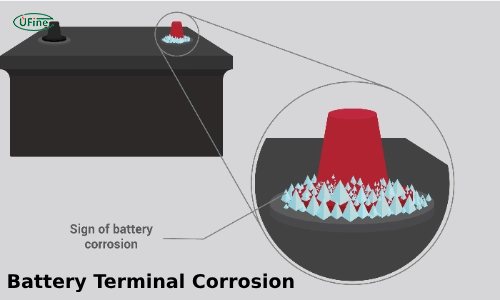
Have you noticed white or green stuff on your car battery? This is battery terminal corrosion. It’s a common problem that can make your car hard to start. This article will explain what causes battery terminal corrosion and how to fix it.
Part 1. Understanding battery terminal corrosion
Battery terminal corrosion is a white, powdery substance buildup on the terminals. This substance can also be green or blue. It forms because of a chemical reaction. When the battery acid leaks and meets the metal of the terminals, it creates corrosion. This happens more often when the battery gets older or is not well-maintained. Corrosion can make it hard for the battery to send power to the car. This can cause starting problems or other electrical issues.
Signs of Battery Terminal Corrosion
You can spot battery terminal corrosion with your eyes. Here are some common signs:
- White, blue, or green powder: Look for this on the battery terminals.
- Dirty or rusted terminals: It could be corrosion if they look dirty or rusty.
- The car has trouble starting: Corrosion can stop the battery from working well, making it hard to start.
- Electrical problems: Things like dim lights or slow power windows can be signs of corrosion.
- Battery warning light: If this light on your dashboard turns on, check for corrosion.
Always check your battery for these signs. Catching corrosion early can save you from more significant problems later.
Part 2. Causes of battery terminal corrosion
Electrolyte Leakage
Electrolyte leakage happens when battery acid seeps out of the battery. This can occur if the battery case cracks or if someone overfills the battery. When the acid leaks, it reaches the battery terminals and reacts with the metal. This reaction forms a white, powdery substance. The leaked electrolyte can cause damage to the battery and the car’s electrical system. It can lead to poor battery performance and difficulty starting the vehicle.
Chemical Reactions
Battery terminal corrosion often results from chemical reactions. When the battery acid, which contains sulfuric acid, touches the metal terminals, it reacts. This reaction can form compounds like copper or zinc sulfate, depending on the terminal material. For example, if the terminals consist of copper, the reaction produces copper sulfate, which appears blue or green. These compounds build up on the terminals and block the flow of electricity.
Overcharging and Undercharging
Overcharging a battery causes the electrolyte to evaporate. This can lead to the buildup of acid residue on the battery terminals. The evaporated acid then condenses on the terminals, creating corrosion. On the other hand, undercharging the battery can also cause problems. A battery receiving insufficient charge can produce lead sulfate crystals, contributing to terminal corrosion. Keeping the battery charged at the right level helps prevent these issues.
Environmental Factors
Environmental conditions play a significant role in battery terminal corrosion. High humidity levels can increase the rate of corrosion. Moisture in the air helps the acid react quickly with the terminal metal. Temperature extremes, both hot and cold, can also accelerate corrosion. Road salt, often used in winter, can splash onto the battery and cause corrosion. Other contaminants, like dirt and oil, can mix with the electrolyte and promote corrosion.
Poor Maintenance
Neglecting battery maintenance leads to many problems, including terminal corrosion. They allow corrosion to build up due to irregular cleaning of the battery terminals. This makes it harder for the battery to work properly. Checking for leaks and securely fastening the battery can prevent acid from spilling onto the terminals. Regular maintenance helps keep corrosion at bay, such as cleaning the terminals with baking soda and water. Always ensure the battery connections are tight and clean to maintain good performance.
Part 3. Preventing battery terminal corrosion
Regular Maintenance
Regular maintenance is critical to preventing battery terminal corrosion. Follow these steps:
- Inspect terminals: Check for any signs of corrosion or buildup regularly.
- Clean terminals: Use baking soda and water to clean terminals.
- Tools and materials: You’ll need a brush or cloth for cleaning, gloves for safety, and a wrench to remove terminals if required.
Proper Charging Techniques
Using the correct charger is crucial for preventing terminal corrosion:
- Choose the suitable charger: Match the charger voltage to your battery’s specifications.
- Avoid overcharging: Overcharging leads to electrolyte evaporation and corrosion.
- Avoid undercharging: Undercharging can cause sulfate crystals to form on terminals.
Protective Measures
Protect your battery terminals with these methods:
- Terminal protectors: Apply terminal protectors or anti-corrosion sprays after cleaning.
- Use of petroleum jelly: Coat terminals with petroleum jelly or specialized greases to prevent moisture buildup.
Environmental Control
Control your battery’s exposure to environmental factors:
- Minimize moisture: Keep battery compartments dry and free of leaks.
- Temperature control: Avoid extreme heat or cold when storing or using the battery.
- Storage tips: If storing the vehicle, disconnect the battery or use a trickle charger to prevent discharge and subsequent corrosion.
Part 4. How to clean battery terminal corrosion?
Cleaning Corroded Terminals
Cleaning corroded battery terminals is essential for maintaining your vehicle’s electrical system. Follow these steps for safe and effective cleaning:
- Safety first – Wear gloves and eye protection to protect yourself from battery acid.
- Disconnect the battery – Remove the negative (black) terminal first, followed by the positive (red) terminal. This prevents accidental short circuits.
- Prepare cleaning solution – Mix baking soda with water to create a paste or solution. This neutralizes acid and aids in corrosion removal.
- Scrub terminals – Use an old toothbrush or battery terminal brush to gently scrub away corrosion and buildup. Make sure to remove all visible corrosion.
- Rinse with water – Use clean water to rinse off the baking soda solution and any remaining corrosion.
- Dry terminals – Thoroughly dry the terminals with a clean cloth to prevent moisture buildup.
Necessary Tools and Safety Precautions
- Tools: Baking soda, water, old toothbrush or battery terminal brush, safety gloves, eye protection.
- Safety Precautions: Avoid contact with battery acid. Work in a well-ventilated area. Keep sparks and flames away from the battery.
Repair and Replacement
Know when it’s time to repair or replace corroded terminals:
- Repair: If corrosion is minor and terminals are salvageable after cleaning, you can apply anti-corrosion spray or terminal protectors.
- Replacement: Consider replacing terminals if corrosion is severe, terminals are damaged, or cleaning doesn’t improve performance.
- Cost considerations: Compare the costs of cleaning supplies versus new terminals. Factor in professional service costs if needed.
- Professional service: Seek a mechanic’s advice for extensive corrosion or terminal damage beyond DIY cleaning.
Part 5. FAQs
-
Does battery terminal corrosion mean the battery is bad?
Battery terminal corrosion itself does not necessarily mean the battery is terrible. It indicates a buildup of residue on the terminals, affecting electrical conductivity. Properly cleaning the terminals can often restore normal function. -
How to fix a corroded battery terminal?
To fix a corroded battery terminal, start by disconnecting the battery. Then, use baking soda and water to clean the terminals with a brush. Rinse with clean water and dry thoroughly. Finally, a small amount of petroleum jelly or terminal protector should be applied to prevent future corrosion. -
Can corroded battery terminals still work?
Corroded battery terminals can still work but may cause poor electrical conductivity and affect battery performance. Regularly cleaning the terminals can help maintain proper function and extend battery life. -
What should you never use to clean a battery terminal?
Never use vinegar or lemon juice to clean battery terminals. These acids are too harsh and can damage the terminals or affect battery performance negatively. Stick to using baking soda mixed with water for safe and effective cleaning.
Related Tags:
More Articles

The Ultimate Guide to Small Size Battery
Learn small battery types, sizes, and applications. Compare lithium, alkaline, and coin cells. Find the best small size battery for your device or project.
Lithium Battery Comparison: Soft Pack Vs. Hard Pack
Compare soft pack vs hard pack lithium battery. Understand structure, safety, energy density, and applications to choose the right battery.
What Size are 18650 Batteries?
Learn exact 18650 battery dimensions (18mm × 65mm), tolerances, and pack configurations. Compare 18650 size with 21700 and others for better battery selection.
The Ultimate Guide to Battery Terminal Clamps
Learn battery terminal clamps, including types, materials, installation, and how to choose the right battery clamp for car and industrial use.
What Is a Lithium-Ion Battery Pack?
Learn what a lithium ion battery pack is, how it works, key components, configurations, and real-world applications for industrial and commercial use.