- Key Takeaways
- Part 1. What is a lithium-air battery?
- Part 2. Lithium air battery working principle
- Part 3. Advantages of lithium air batteries
- Part 4. Key challenges limiting lithium air batteries
- Part 5. Applications: Where lithium air batteries fit
- Part 6. Lithium-air vs lithium-ion vs lead-acid
- Part 7. Lithium air battery R&D progress and industry trends
- Part 8. Lithium air battery selection guide for engineers
- Part 9. FAQs about lithium air battery
Lithium-air batteries (also called Li-air batteries or lithium ion air batteries) are one of the most promising next-generation energy storage technologies. They offer extremely high theoretical energy density, far beyond traditional lithium-ion systems.
But in real-world engineering, performance, stability, and lifespan still limit their commercial use.
This guide explains the lithium air battery working principle, compares it with other technologies, and helps you understand when (and if) it makes sense to consider Li-air systems.
Key Takeaways
- Lithium air batteries can reach theoretical energy densities up to ~5,000 Wh/kg, but practical values are much lower today.
- The lithium air battery working principle relies on oxygen from air, reducing weight but increasing system complexity.
- The biggest barriers are cycle life, electrolyte stability, and sensitivity to moisture/CO₂.
- Compared to lithium-ion, Li-air batteries are not yet ready for commercial deployment in most applications.
- They are best viewed as a future technology for ultra-lightweight energy storage, especially in EV and aerospace sectors.
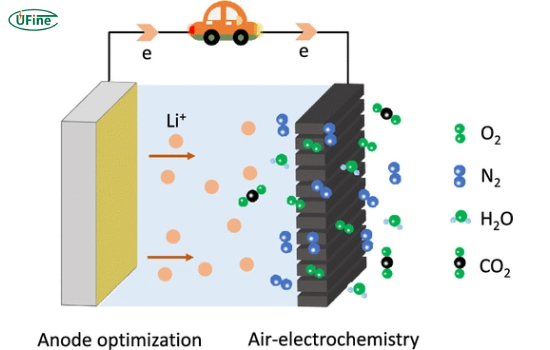
Part 1. What is a lithium-air battery?
A lithium-air battery is a type of electrochemical battery that uses:
- Lithium metal as the anode
- Oxygen from ambient air as the cathode reactant
Unlike sealed batteries, Li-air systems rely on external oxygen, which significantly reduces weight and increases theoretical capacity.
👉 For a deeper comparison with current mainstream tech, see: Lithium-air vs lithium-ion battery
Key concept
During discharge, lithium reacts with oxygen to form:
- Lithium peroxide (Li₂O₂)
- or Lithium oxide (Li₂O)
This reaction is the source of its extremely high energy density.
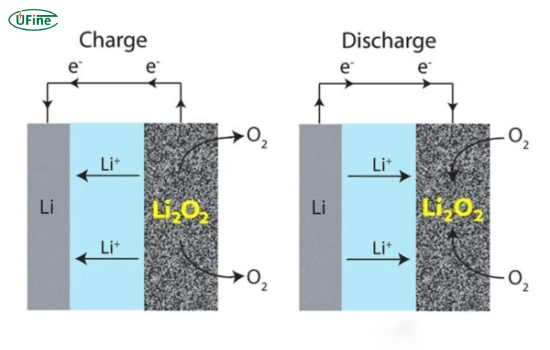
Part 2. Lithium air battery working principle
Understanding the lithium air battery working principle is critical for evaluating its feasibility.
Discharge process
- Lithium metal releases electrons (oxidation)
- Lithium ions move through the electrolyte
- Oxygen enters the porous cathode
- Lithium reacts with oxygen → forms Li₂O₂ / Li₂O
Charge process
- The reaction reverses
- Lithium compounds decompose
- Oxygen is released back into the air
Engineering note
The cathode must be:
- Porous (to allow oxygen flow)
- Chemically stable (to resist side reactions)
This requirement makes Li-air design far more complex than sealed batteries.
Part 3. Advantages of lithium air batteries
-
Ultra-high energy density
- Theoretical: up to ~5,000 Wh/kg
- Lithium-ion: typically 150–250 Wh/kg
- This makes lithium air batteries highly attractive for:
- Long-range electric vehicles
- Aerospace systems
-
Lightweight system design
- Because oxygen comes from air
- No need to store oxidizer internally
- Lower total battery mass
-
Potential material sustainability
- Li-air batteries may use fewer heavy metals
- More abundant cathode inputs
Part 4. Key challenges limiting lithium air batteries
Despite strong potential, lithium air batteries are not yet commercially viable.
-
Poor cycle life
- Often limited to <200 cycles
- Cathode clogging from reaction products
-
Electrolyte instability
- Electrolytes can react with oxygen radicals
- Electrolytes can decompose during cycling
- This reduces efficiency and lifespan
-
Air contamination issues
- Real air contains moisture (H₂O)
- Real air contains carbon dioxide (CO₂)
- These cause side reactions
- Carbonate formation
- Rapid degradation
-
Low round-trip efficiency
- Energy loss during charge/discharge
- High overpotential
Part 5. Applications: Where lithium air batteries fit
-
Electric vehicles (future use)
- Potential for 2–5× range increase
- Still under research due to durability limits
-
Aerospace and drones
- Weight-sensitive applications
- High energy density is critical
-
Grid-scale storage (long-term)
- Possible use in low-weight transportable storage
- Not yet practical for daily cycling
Part 6. Lithium-air vs lithium-ion vs lead-acid
| Feature | Lithium Air Battery | Lithium-Ion Battery | Lead-Acid Battery |
|---|---|---|---|
| Energy Density | Up to 5,000 Wh/kg (theoretical) | 150–250 Wh/kg | 30–50 Wh/kg |
| Cycle Life | 100–200 cycles | 500–1,500 cycles | 500–1,000 cycles |
| Efficiency | Low–moderate | High (>90%) | Moderate |
| Weight | Very light | Medium | Heavy |
| Maturity | Experimental | Commercial | Mature |
Practical takeaway
- Lithium-ion = best for current products
- Lead-acid = low-cost, robust
- Lithium-air = future, high-risk/high-reward
Part 7. Lithium air battery R&D progress and industry trends
Current research focuses on:
-
Advanced cathode materials
- Catalysts to improve oxygen reactions
- Nanostructured porous electrodes
-
Stable electrolytes
- Solid-state electrolytes
- Non-aqueous systems
-
Closed-system Li-air designs
- Filtering CO₂ and moisture
- Improving real-world stability
Key research players
- IBM Research
- A123 Systems
- Leading university labs in electrochemistry
👉 For broader battery technology trends, see: IEA Global EV Outlook
Part 8. Lithium air battery selection guide for engineers
Before considering lithium air batteries, evaluate:
When NOT to use Li-air
- Commercial products (today)
- High cycle applications
- Harsh outdoor environments
When to monitor Li-air
- Next-gen EV platforms
- Aerospace projects
- Ultra-lightweight R&D systems
👉 If you need ready-to-deploy lithium battery solutions, explore: Custom lithium battery solutions
Part 9. FAQs about lithium air battery
What is a lithium air battery?
A lithium air battery is a rechargeable battery that uses lithium metal and oxygen from air to generate electricity, enabling very high theoretical energy density.
How does a lithium air battery work?
The lithium air battery working principle involves lithium reacting with oxygen during discharge and reversing the reaction during charging.
Why are lithium air batteries not widely used?
They suffer from short cycle life, electrolyte instability, and sensitivity to air contaminants, which limit real-world use.
Are lithium air batteries better than lithium-ion?
In theory, yes (higher energy density). In practice, lithium-ion batteries are far more reliable and commercially viable.
What is the future of lithium air batteries?
They are considered a long-term breakthrough technology, but widespread adoption will depend on solving durability and efficiency challenges.
Related Tags:
More Articles

What is the KC Certification of Lithium Battery?
Understand KC certification for lithium batteries, including requirements, testing process, kc battery compliance, and how it compares with CE and UL.
How to Dispose of Swollen Lithium Polymer Battery?
Safely dispose of swollen lithium polymer (LiPo) batteries. Learn proper storage, handling, and recycling steps to prevent fire risks.
Battery Technology: How to Spot Weld Lithium Batteries?
Learn how to spot weld batteries using a battery spot welder. Includes setup, welding parameters, nickel strips, and tips for lithium battery packs.
Best Military Battery: Top 10 Batteries for Military Applications in 2026
Explore the top 10 military batteries for durable, reliable performance. Compare LiFePO4, AGM, and lead-acid options built for extreme conditions.
Battery Solutions Guides: Definitions, Components, Types, and More
Learn what a battery solution is, its components, types, and real-world uses. Practical guide for selecting the right battery solution for your application.