Batteries power almost every modern device. Many people ask how do batteries work and what is the battery working principle behind them.
A battery converts chemical energy into electrical energy. This electricity powers devices such as smartphones, electric vehicles, and industrial equipment. Understanding how batteries work helps improve battery life, system performance, and selection decisions.
Key Takeaways
- A battery works through redox reactions that convert chemical energy into electricity.
- During discharge, electrons flow through the circuit while ions move inside the battery.
- Charging reverses the chemical reaction and restores energy.
- Battery performance depends on temperature, discharge rate, and cycle life.
- Different battery types use different materials but follow the same working principle.
Part 1. How does a battery work?
1 Battery working principle
The battery working principle is based on electrochemical reactions.
A battery consists of:
- Anode (negative electrode)
- Cathode (positive electrode)
- Electrolyte (ion transfer medium)
When connected to a device:
- Electrons flow through the external circuit
- Ions move inside the battery through the electrolyte
This process generates electrical energy, which explains how a battery works.
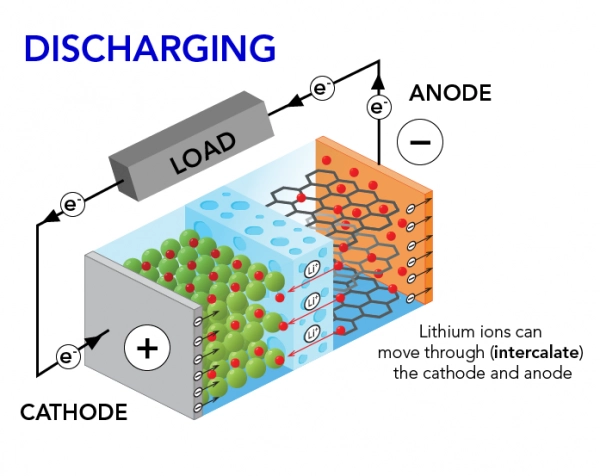
2 Battery discharging process
During discharge:
- The anode releases electrons
- Electrons flow through the external circuit and power the device
- Lithium ions move from anode to cathode through the electrolyte
This synchronized movement of electrons and ions produces usable electricity.
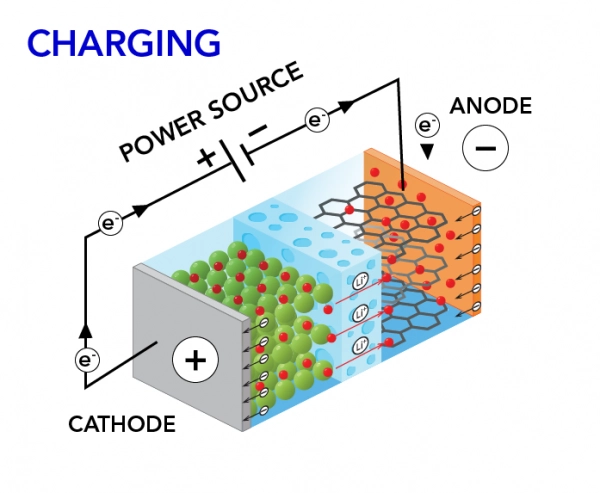
3 How does charging a battery work?
Charging reverses the process.
- External power pushes electrons back to the anode
- Lithium ions move from cathode to anode
- Chemical energy is restored inside the battery
This explains how does charging a battery work in practical systems.
Part 2. Battery chemical reaction and components
1 Types of batteries
Batteries fall into two main categories:
| Type | Description | Applications |
|---|---|---|
| Primary batteries | Non-rechargeable | Remote controls, sensors |
| Secondary batteries | Rechargeable | EVs, energy storage |
For more details, see lithium polymer battery guide
2 Chemical reactions in batteries
All batteries operate through redox reactions:
- Oxidation at the anode releases electrons
- Reduction at the cathode accepts electrons
- Non-rechargeable batteries
- One-way reaction
- Cannot be recharged
- Rechargeable batteries
- Reversible reactions
- Support multiple charge cycles
Reference: electrochemistry basics
3 Battery anode, cathode, and electrolyte
| Component | Function |
|---|---|
| Anode | Releases electrons |
| Cathode | Accepts electrons |
| Electrolyte | Transfers ions |
Material selection determines:
- Energy density
- Safety
- Lifespan
Part 3. Battery performance factors
1 Temperature effects on batteries
- High temperature accelerates degradation
- Low temperature reduces capacity
2 Discharge rate and battery efficiency
- Higher discharge rate reduces usable capacity
- Increases internal heat and stress
3 Battery cycle life
- Each charge cycle reduces capacity
- Cycle life varies by chemistry
Related reading: battery cycle life guide
4 Battery storage conditions
Best practices:
- Store at 30–60% charge
- Keep in a cool and dry environment
Part 4. Battery applications and selection guide
Understanding what batteries do helps match the right battery to each use case.
| Application | Requirement | Battery Type |
|---|---|---|
| Electric vehicles | High energy density | NMC / NCA |
| Solar storage | Long cycle life | LiFePO₄ |
| Portable devices | Lightweight | Li-ion / LiPo |
| Backup power | Stability | LFP / Lead-acid |
Part 5. How do batteries work FAQs
How do batteries work in simple terms?
Batteries convert chemical energy into electrical energy. Electrons flow through a circuit while ions move inside the battery.
What is the function of a battery?
The function of a battery is to store energy and release it as electricity when needed.
Why do batteries go flat?
Batteries go flat when the internal chemical materials are depleted and can no longer produce electrical energy.
Are batteries AC or DC?
Batteries provide direct current (DC), meaning electricity flows in one direction.
What is inside a battery?
A battery contains an anode, cathode, and electrolyte, which enable chemical reactions that generate electricity.
Related Tags:
More Articles

The Ultimate Guide to Small Size Battery
Learn small battery types, sizes, and applications. Compare lithium, alkaline, and coin cells. Find the best small size battery for your device or project.
Lithium Battery Comparison: Soft Pack Vs. Hard Pack
Compare soft pack vs hard pack lithium battery. Understand structure, safety, energy density, and applications to choose the right battery.
What Size are 18650 Batteries?
Learn exact 18650 battery dimensions (18mm × 65mm), tolerances, and pack configurations. Compare 18650 size with 21700 and others for better battery selection.
The Ultimate Guide to Battery Terminal Clamps
Learn battery terminal clamps, including types, materials, installation, and how to choose the right battery clamp for car and industrial use.
What Is a Lithium-Ion Battery Pack?
Learn what a lithium ion battery pack is, how it works, key components, configurations, and real-world applications for industrial and commercial use.