- Quick Answer
- Part 1. Before lithium-ion: why a new battery was needed
- Part 2. Who invented the lithium-ion battery?
- Part 3. What was the first lithium-ion battery?
- Part 4. A timeline of lithium-ion battery development
- Part 5. Why lithium was always the ideal—but dangerous—choice
- Part 6. The 3 scientific breakthroughs that made lithium-ion possible
- Part 7. From laboratory to factory: why sony succeeded first
- Part 8. The evolution of lithium-ion battery materials
- Part 9. Why lithium-ion batteries enabled entire industries
- Part 10. The limits of lithium-ion
- Part 11. Where lithium-ion technology is heading next
- Part 12. Final perspective
- Part 13. FAQs
Lithium-ion batteries did not become dominant overnight. Their rise was slow, complex, and deeply tied to advances in materials science, global energy demand, and the evolution of consumer electronics.
Today, lithium-ion technology sits at the center of electric vehicles, renewable energy storage, and portable electronics. But behind this success lies decades of trial, failure, and scientific debate. Understanding how lithium-ion batteries evolved—and why certain design choices won over others— offers valuable insight into where battery technology is heading next.
Quick Answer
Lithium-ion batteries emerged in the 1980s, when researchers solved a fundamental safety problem associated with metallic lithium.
The first commercially viable lithium-ion battery was developed in 1985 by Akira Yoshino, and the first mass-produced lithium-ion battery entered the market in 1991, released by Sony.
This innovation transformed rechargeable batteries from bulky, low-energy systems into compact, high-performance power sources.
Part 1. Before lithium-ion: why a new battery was needed
To understand why lithium-ion batteries were such a breakthrough, we need a bit of context.
Before the 1980s, rechargeable batteries mainly relied on technologies like:
- Lead-acid batteries
- Nickel-cadmium (Ni-Cd) batteries
- Nickel-metal hydride (NiMH) batteries
While useful, these batteries had clear limitations. They were heavy, had relatively low energy density, and often suffered from memory effects or safety concerns. As portable electronics began to emerge, researchers needed a lighter, more energy-dense solution.
Lithium, being the lightest metal and highly reactive, looked like the perfect candidate.
Part 2. Who invented the lithium-ion battery?
The invention of the lithium-ion battery was not the work of a single person. Instead, it was the result of decades of research by several key scientists.
Stanley Whittingham: The First Breakthrough (1970s)
In the early 1970s, Stanley Whittingham, working at Exxon, developed one of the first rechargeable lithium batteries. His design used:
- A titanium disulfide cathode
- A metallic lithium anode
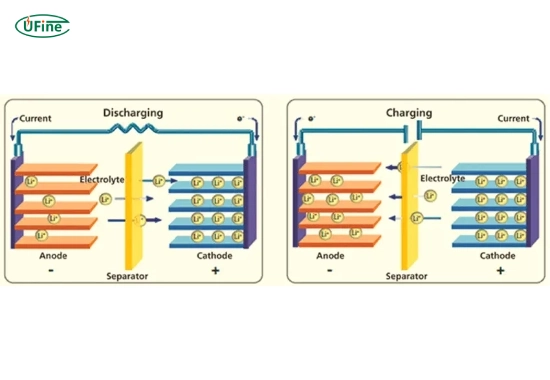
More importantly, Whittingham introduced the concept of intercalation, where lithium ions move in and out of a host material without destroying its structure. This idea became the backbone of modern lithium-ion batteries.
However, metallic lithium caused safety issues, including dendrite formation and short circuits. The concept was promising—but not yet safe enough.
John B. Goodenough: Raising the Voltage (1980)
The next major leap came in 1980, when John B. Goodenough discovered that lithium cobalt oxide (LiCoO₂) could be used as a cathode material.
This discovery dramatically increased battery voltage and energy density, making lithium-based batteries far more practical. In many ways, this was the turning point that made lithium-ion batteries commercially possible.
Goodenough’s contribution later earned him a share of the 2019 Nobel Prize in Chemistry, alongside Whittingham and Yoshino
Akira Yoshino: Making It Safe and Commercial (1985)
While Whittingham and Goodenough laid the scientific foundation, it was Akira Yoshino who solved the final piece of the puzzle.
In 1985, Yoshino replaced metallic lithium with a carbon-based anode, allowing lithium ions—rather than lithium metal—to move during charging and discharging. This design dramatically improved safety and stability.
Yoshino’s battery is widely recognized as the first true lithium-ion battery, suitable for real-world use.
Part 3. What was the first lithium-ion battery?
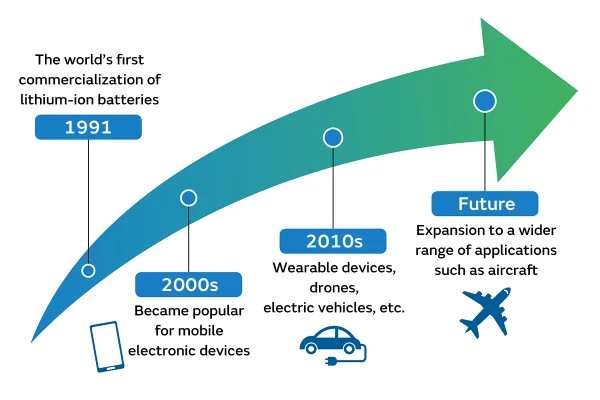
The first practical lithium-ion battery appeared in 1985, but it wasn’t until 1991 that lithium-ion batteries reached the market.
That year, Sony released the world’s first commercial lithium-ion battery, initially used in portable electronics such as camcorders. This launch changed everything.
For the first time, consumers had access to a rechargeable battery that was:
- Lightweight
- High in energy density
- Relatively safe
- Long-lasting
From that point on, lithium-ion batteries rapidly replaced older rechargeable technologies.
Part 4. A timeline of lithium-ion battery development
Rather than listing every single event, it’s more helpful to look at lithium-ion history in key phases.
1970s–1980s: Scientific Foundations
- Whittingham introduces lithium intercalation
- Goodenough discovers lithium cobalt oxide
- Yoshino develops the first safe lithium-ion design
1990s: Commercialization
- 1991: Sony launches the first commercial lithium-ion battery
- Lithium-ion batteries begin replacing Ni-Cd and NiMH in electronics
- Early safety incidents drive improvements in battery management
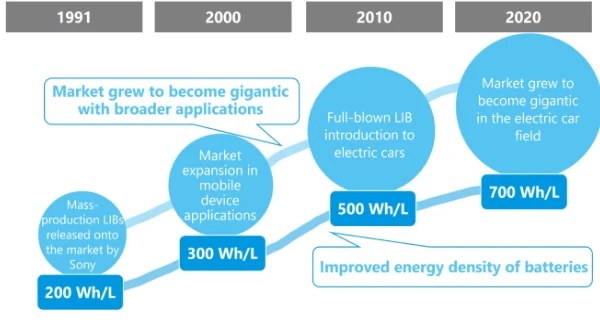
2000s: Performance Improvements
- Higher energy density materials
- Better electrolytes and separators
- Growing use in laptops, power tools, and medical devices
2010s–Present: Electric Vehicles and Energy Storage
- Rapid adoption in EVs
- Development of lithium iron phosphate (LiFePO₄)
- Expansion into grid-scale energy storage
Part 5. Why lithium was always the ideal—but dangerous—choice
From a theoretical standpoint, lithium was always the perfect battery material.
It is:
- The lightest metal in the periodic table
- Extremely electropositive, enabling high cell voltage
- Capable of storing large amounts of energy per unit mass
However, early lithium batteries used metallic lithium, which introduced a critical flaw. During repeated charging cycles, lithium tends to form dendrites—needle-like structures that can pierce separators and cause internal short circuits.
For years, lithium batteries promised high energy density but failed in real-world safety.
The lithium-ion battery was born not from discovering lithium—but from learning how to use lithium without metallic lithium.
Part 6. The 3 scientific breakthroughs that made lithium-ion possible
Lithium-ion batteries exist because three independent scientific ideas eventually converged.
Intercalation: The Concept That Changed Everything
In the 1970s, Stanley Whittingham demonstrated that lithium ions could be inserted into layered materials without permanently altering their structure. This process—called intercalation—allowed energy storage without consuming lithium metal during operation.
Although Whittingham’s early batteries still used metallic lithium, intercalation proved that lithium could move reversibly inside solid hosts.
This idea became the backbone of all modern lithium-ion batteries.
High-Voltage Cathodes: Unlocking Practical Energy Density
Intercalation alone was not enough. Early cathode materials produced insufficient voltage.
That changed in 1980, when John B. Goodenough identified lithium cobalt oxide (LiCoO₂) as a stable, high-voltage cathode material. This single discovery effectively doubled the energy density achievable in rechargeable batteries.
Without LiCoO₂, lithium-ion batteries would never have reached commercial relevance.
Goodenough’s work later earned global recognition, including the 2019 Nobel Prize in Chemistry
Removing Metallic Lithium: The Final Safety Barrier
The last—and arguably most important—step came in 1985, when Akira Yoshino replaced metallic lithium with a carbon-based anode.
Instead of plating lithium metal during charging, Yoshino’s design allowed lithium ions to shuttle safely between electrodes. This eliminated dendrite growth and dramatically improved cycle life and safety.
At this point, the modern lithium-ion battery architecture was complete.
Part 7. From laboratory to factory: why sony succeeded first
Many companies understood the promise of lithium-ion batteries, but Sony was the first to commercialize them successfully.
Sony’s advantage was not only chemistry—it was manufacturing discipline:
- Strict quality control
- Early adoption of battery protection circuits
- Conservative energy density targets to prioritize safety
In 1991, Sony introduced the first commercial lithium-ion battery, initially for portable electronics. Within a few years, lithium-ion technology displaced nickel-based batteries across the consumer market.
This shift quietly reshaped the electronics industry.
Key Milestones in the Invention of Lithium-Ion Batteries
| Year | Scientist / Organization | Key Breakthrough | Why It Mattered |
|---|---|---|---|
| 1973–1976 | Stanley Whittingham (Exxon) | Introduced lithium-ion intercalation using TiS₂ | Proved lithium ions could move reversibly in solid materials |
| 1980 | John B. Goodenough (Oxford) | Discovered lithium cobalt oxide (LiCoO₂) cathode | Enabled high-voltage, high-energy rechargeable batteries |
| 1985 | Akira Yoshino | Replaced metallic lithium with carbon anode | Solved safety issues and enabled real-world use |
| 1991 | Sony | First commercial lithium-ion battery | Marked the start of mass-market adoption |
| Late 1990s | Multiple manufacturers | Battery management & safety systems | Improved reliability and reduced failure risks |
Part 8. The evolution of lithium-ion battery materials
Once lithium-ion batteries entered mass production, innovation shifted from invention to optimization.
Cathode Diversification
While lithium cobalt oxide dominated early designs, it was expensive and thermally sensitive. This led to the development of:
- Lithium manganese oxide (LMO) for lower cost
- Lithium iron phosphate (LiFePO₄) for thermal stability and safety
- Nickel-rich chemistries (NCA, NCM) for higher energy density
Each chemistry represents a trade-off between energy density, safety, cost, and lifespan.
Anode and Electrolyte Improvements
Graphite remained the dominant anode material, but refinements in particle morphology and electrolyte additives greatly improved:
- Cycle life
- Fast-charging capability
- Low-temperature performance
These incremental improvements enabled lithium-ion batteries to scale from phones to electric vehicles.
Part 9. Why lithium-ion batteries enabled entire industries
Lithium-ion batteries didn’t just replace older batteries—they created new markets.
They made possible:
- Ultra-thin smartphones and laptops
- Long-range electric vehicles
- Grid-scale renewable energy storage
- High-power tools and drones
No other rechargeable battery chemistry has matched lithium-ion’s balance of energy density, scalability, and manufacturability.
Part 10. The limits of lithium-ion
Despite their success, lithium-ion batteries are not perfect.
Key challenges include:
- Thermal runaway under extreme abuse
- Dependence on critical raw materials
- Performance degradation over time
These limitations are driving research into next-generation solutions, rather than signaling the end of lithium-ion dominance.
Part 11. Where lithium-ion technology is heading next
Solid-State Architectures
Replacing liquid electrolytes with solid materials could dramatically improve safety and energy density, though large-scale manufacturing remains challenging.
Sustainability and Recycling
Future growth depends on improved recycling efficiency and reduced reliance on scarce materials.
System-Level Innovation
Battery packs, management systems, and thermal design are becoming just as important as cell chemistry itself.
Part 12. Final perspective
Lithium-ion batteries represent one of the most impactful technological achievements of the late 20th century. Their invention required not a single breakthrough, but the careful alignment of chemistry, materials science, and manufacturing discipline.
While new technologies may eventually complement or replace them, lithium-ion batteries will remain the foundation of modern energy storage for years to come.
Part 13. FAQs
Why did early lithium batteries fail before lithium-ion technology succeeded?
Early lithium batteries used metallic lithium, which is highly reactive. During charging, lithium metal can form dendrites that grow inside the battery and eventually cause internal short circuits. This made early designs unsafe and unreliable. Lithium-ion batteries solved this problem by using lithium ions stored in stable host materials, eliminating metallic lithium and greatly improving safety and lifespan.
What makes lithium-ion batteries fundamentally different from other rechargeable batteries?
Lithium-ion batteries rely on reversible ion intercalation, rather than irreversible chemical reactions. This allows them to achieve much higher energy density, higher operating voltage, and longer cycle life than technologies like lead-acid or nickel-based batteries. As a result, they are better suited for compact electronics and scalable energy systems.
Why are there so many lithium-ion chemistries if the technology was already invented?
Different lithium-ion chemistries exist because no single battery design optimizes everything at once. Some chemistries prioritize energy density, others focus on safety, cost, or lifespan. For example, electric vehicles, consumer electronics, and energy storage systems all have different performance requirements, leading to multiple optimized lithium-ion variants.
What limits the lifespan of lithium-ion batteries over time?
Lithium-ion batteries degrade due to gradual loss of active lithium, growth of internal resistance, and stress from heat and charging behavior. Even under ideal conditions, these chemical and structural changes accumulate over time, making capacity loss unavoidable. Proper temperature control and charging practices can slow—but not stop—this process.
How close are lithium-ion batteries to their practical performance limits?
Conventional lithium-ion batteries are approaching their practical limits in terms of energy density and safety. Most recent improvements are incremental rather than revolutionary. Significant future gains will likely come from new architectures, such as solid-state designs, rather than from traditional lithium-ion chemistry alone.
Related Tags:
More Articles
What is the Lithium Battery Charging Cycle?
Learn what a lithium ion battery charge cycle is, how cycle count affects lifespan, and proven ways to extend battery cycle life in real applications.
What is a 9V Battery? A Comprehensive Guide
Complete 9V battery guide: internal structure, alkaline vs lithium vs rechargeable, lifespan, applications, testing, and safe disposal. Updated for 2026.
Lead Acid vs. Lithium Ion Batteries: A Complete Comparison
Compare lead acid vs lithium-ion batteries in safety, cost per kWh, energy density, size, lifespan, and applications. Selection guide for engineers and buyers.
Is Your Lithium Battery Secretly Failing?
Find out the common causes of lithium battery failure and simple strategies to keep your devices running longer.
Learn how to charge a drone battery correctly. This guide covers battery types, chargers, safe charging steps, and best practices to extend battery life.