- Key Takeaways
- Part 1. Battery types
- Part 2. Battery components (internal structure explained)
- Part 3. Battery performance metrics
- Part 4. Battery charging and discharging terminology
- Part 5. Battery connections (series vs parallel)
- Part 6. Battery safety and maintenance terminology
- Part 7. Battery terminology FAQs
Batteries power everything—from small electronics to EVs and industrial systems. If you work with batteries, understanding battery terminology is critical for design, selection, and procurement decisions.
This guide explains key battery vocabulary, including battery types, internal structure, performance metrics, and safety terms. It is written for engineers, buyers, and technical teams who need clear and practical knowledge.
Key Takeaways
- Battery terminology helps you make better decisions in design, sourcing, and system integration.
- The battery internal diagram (anode, cathode, electrolyte, separator) explains how energy flows.
- Key metrics like capacity, voltage, and cycle life directly impact performance and cost.
- Charging methods and BMS control are critical for safety and lifespan.
- Series vs parallel connections determine system voltage and capacity.
Part 1. Battery types
Understanding battery types is the first step in mastering battery terminology.
| Battery Type | Key Features | Typical Applications |
|---|---|---|
| Lead-Acid | Low cost, reliable | UPS, backup systems |
| Lithium-Ion | High energy density, lightweight | EVs, portable devices |
| NiCd | Durable, works in harsh conditions | Aviation, industrial |
| NiMH | Safer, eco-friendly | Hybrid vehicles |
| Alkaline | Long shelf life | Consumer electronics |
| Zinc-Carbon | Low cost | Low-drain devices |
👉 For most modern applications, lithium-ion batteries dominate due to efficiency and lifespan.
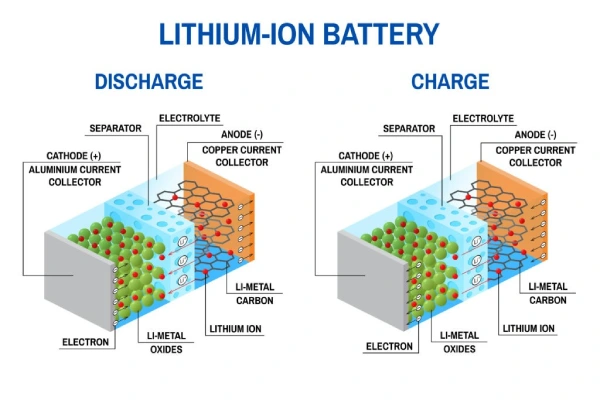
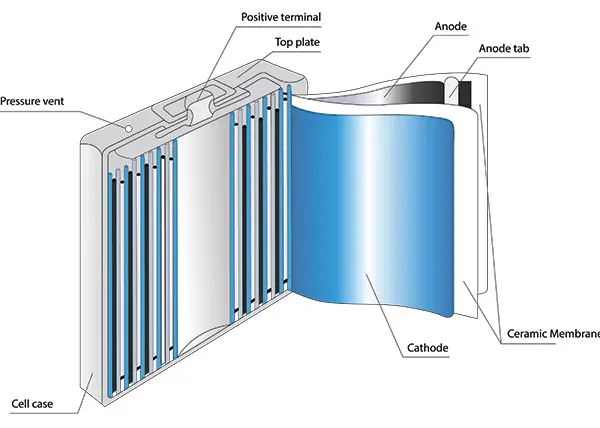
Part 2. Battery components (internal structure explained)
To understand a battery internal diagram, focus on these core components:
- Anode (Negative Electrode)
- Releases electrons during discharge
- Typically made of graphite in lithium batteries
- Cathode (Positive Electrode)
- Accepts electrons
- Determines battery voltage and chemistry
- Electrolyte
- Transfers ions between electrodes
- Can be liquid, gel, or solid
- Separator
- Prevents short circuits
- Allows ion flow
- Terminals
- External connection points
- Casing
- Protects internal structure
- Provides mechanical strength
- Cell
- Basic unit of a battery
- Multiple cells form a battery pack
👉 Learn more about energy units here: Watts and Watt-hours explained
Part 3. Battery performance metrics
These are the most important battery terms for evaluation and comparison.
- Capacity (Ah / mAh)
- Total charge stored
- Higher = longer runtime
- Voltage (V)
- Electrical potential difference
- Determines system compatibility
- Current (A)
- Flow of electric charge
- Energy Density (Wh/kg or Wh/L)
- Energy per weight or volume
- Critical for portable devices
- Power Density (W/kg)
- How fast energy can be delivered
- Important for high-load systems
- Cycle Life
- Number of charge/discharge cycles
- Key factor in total cost of ownership
- Internal Resistance
- Affects efficiency and heat generation
- C-Rate
- Charge/discharge speed relative to capacity
- Efficiency
- Energy output vs input
- Self-Discharge Rate
- Energy loss during storage
👉 For deeper understanding of temperature effects: Battery performance at extreme temperatures
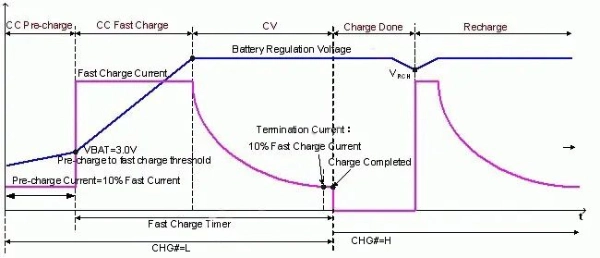
Part 4. Battery charging and discharging terminology
Charging behavior directly impacts battery life and safety.
- Charging
- Restores battery energy
- Requires controlled voltage and current
- Discharging
- Releases stored energy
- Must stay within safe limits
- Overcharging
- Causes heat and degradation
- Can lead to failure
- Deep Discharge
- Damages battery chemistry
- Reduces lifespan
- Fast Charging
- Reduces charging time
- May increase thermal stress
- Trickle Charging
- Maintains charge over time
- Float Charging
- Keeps battery at constant voltage (common in backup systems)
- Battery Management System (BMS)
- Monitors voltage, temperature, current
- Prevents unsafe conditions
- Essential for lithium battery packs
Part 5. Battery connections (series vs parallel)
Battery configuration determines system output.
- Series Connection
- Increases voltage
- Capacity stays the same
- Parallel Connection
- Increases capacity
- Voltage stays the same
- Series-Parallel
- Combines both
- Used in EVs and energy storage systems
👉 Correct configuration is critical for system design and safety.
Part 6. Battery safety and maintenance terminology
Safety-related battery vocabulary is essential for real-world applications.
- Thermal Runaway
- Uncontrolled temperature rise
- Can cause fire or explosion
- Overheating
- Reduces lifespan
- Needs cooling and ventilation
- Short Circuit
- Direct connection of terminals
- Causes high current and damage
- Venting
- Releases internal pressure
- Prevents explosion
- Battery Degradation
- Gradual capacity loss
- Influenced by cycles, heat, and usage
- Cell Balancing
- Ensures equal charge across cells
- Improves performance and lifespan
- State of Charge (SoC)
- Remaining battery capacity (%)
- State of Health (SoH)
- Overall battery condition vs new
👉 For safety standards and best practices, refer to:
Part 7. Battery terminology FAQs
What is the most important battery terminology for beginners?
Start with capacity, voltage, cycle life, and energy density. These define performance and application fit.
What does a battery internal diagram show?
It shows key components like anode, cathode, electrolyte, and separator, explaining how energy flows inside a battery.
Which battery type is best for industrial use?
Lithium-ion batteries are widely used due to high efficiency, long cycle life, and low maintenance.
How can I extend battery lifespan?
Avoid overcharging, deep discharge, and high temperatures. Use a reliable BMS.
What is the difference between series and parallel connections?
Series → higher voltage. Parallel → higher capacity.
Related Tags:
More Articles

Battery Nominal Voltage: What You Need to Know
Learn what battery nominal voltage is, how it is determined, why it matters for performance and safety, and get charts, comparisons, and selection guidance.
The Complete Guide to Lithium Ion Battery Voltage Chart
Lithium battery voltage chart explained: compare 3.2V LFP, 3.7V NCM, and 4.2V Li-ion cells. Learn voltage range, SOC chart, and how to choose the right battery.
A Comprehensive Guide to Lawn Mower Battery
Learn lawn mower battery types, lifespan, and how to choose the right battery. Covers voltage, capacity, maintenance, and common issues.
What Is a Smart Battery? A Comprehensive Guide
Learn what a smart battery is, how smart batteries work, and why they improve safety, lifespan, and performance. Includes comparison, applications, and tips.
4680 Battery Cell vs 18650: A Detailed Comparison
Compare 4680 vs 18650 battery cells in terms of capacity, size, energy density, and applications. Learn which battery type is best for EVs and industrial use.